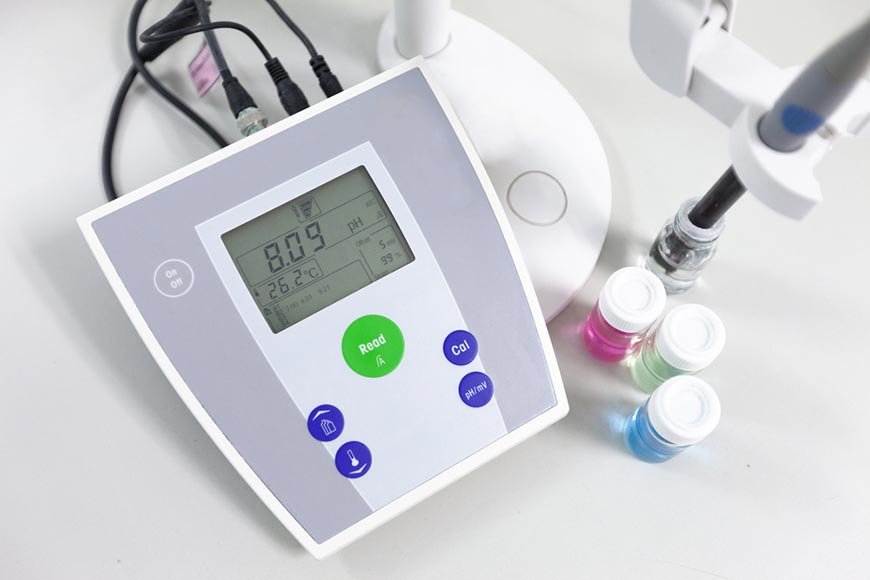
Accurate calibration ensures reliable results in laboratories and industrial processes. Instruments like a pH Calibrating Buffer provide a stable reference for measurements, preventing drift caused by environmental changes or aging sensors. Correct calibration minimizes errors, safeguards sensitive solutions, and reduces waste in experiments. Laboratories and manufacturers rely on precise instruments to achieve reproducible outcomes, maintain chemical integrity, and uphold safety standards. Consistent calibration practices form the backbone of stable solution handling and trustworthy experimental or industrial results.
Solution Stability
Solution stability refers to the ability of a chemical mixture to maintain its properties over time. Factors such as temperature fluctuations, pH drift, and contamination can compromise this stability. Regular calibration ensures that instruments accurately detect changes, preventing unnoticed deviations. Stable solutions enhance reaction predictability, improve product quality, and reduce experimental errors. Monitoring and adjusting instruments provide accurate data for decision-making. By maintaining solution consistency, laboratories and industrial operations save resources and achieve repeatable results with confidence in both short-term and long-term applications.
Key Factors Affecting Solution Stability
Several elements influence solution stability, including:
- Temperature changes that accelerate chemical reactions
- Light exposure causing breakdown of sensitive compounds
- Contaminants introduced during handling
- Measurement errors from instruments
- Shifts in pH, concentration, or conductivity
Monitoring these factors with calibrated instruments ensures solutions remain within desired specifications. Routine checks allow professionals to detect deviations early, maintain chemical integrity, and reduce the risk of experimental failure or production inconsistencies. Calibration is essential to manage these variables effectively.
The Role of Calibration in Maintaining Accuracy
Calibration aligns measurement instruments with known standards to deliver accurate readings. Devices such as pH meters, conductivity probes, and titration equipment require regular calibration to prevent drift. This process corrects errors from sensor aging or environmental changes. Accurate readings maintain solution integrity, improve reproducibility, and safeguard experiments. Industries depend on calibration to uphold quality standards and comply with regulations. Reliable instruments also allow personnel to make precise adjustments in real-time, ensuring processes run smoothly and results remain consistent across repeated measurements or production cycles.
Benefits of Using Standardized Calibration Tools
Standardized calibration tools, such as pH buffers and conductivity solutions, provide reliable references that ensure reproducible measurements. Using these tools offers several advantages:
- Enhanced precision when monitoring chemical reactions
- Reduced risk of measurement errors
- Increased confidence in experimental and production data
- Compliance with industry regulations
- Extended lifespan of sensitive analytical instruments
These benefits collectively strengthen solution stability. Consistently employing standardized tools allows laboratories and industries to maintain accurate measurements across diverse applications without risking chemical degradation or inconsistent results.
Practical Applications Across Industries
Calibration has a significant impact in various sectors:
- Pharmaceuticals: Maintains drug potency and formula consistency
- Food and Beverage: Ensures safety and product quality
- Chemical Manufacturing: Stabilizes reaction conditions and outputs
- Environmental Testing: Provides reliable water, soil, and air analysis
- Biotechnology: Supports controlled conditions for cells and enzymes
Industries relying on precise chemical measurements gain efficiency, reduce waste, and improve consistency. Accurate calibration ensures solutions behave predictably, safeguarding both product quality and safety for end-users.
Common Calibration Practices to Improve Solution Stability
Adopting proper calibration routines is critical for stability:
- Verify instruments with certified standards regularly
- Maintain detailed records of calibration results and schedules
- Adjust devices promptly if deviations are detected
- Train staff for consistent calibration procedures
- Store calibration materials in controlled environments
Following these practices ensures long-term stability of solutions. Systematic calibration routines prevent unnoticed errors, maintain chemical integrity, and enhance reproducibility across experiments or industrial processes. These steps support reliable decision-making and operational efficiency.
Challenges in Achieving Optimal Solution Stability
Despite calibration, achieving complete stability can be challenging. Factors such as extreme environmental conditions, highly reactive solutions, and instrument limitations can affect outcomes. Mitigation requires monitoring, timely recalibration, and understanding solution-specific sensitivities. Controlled storage conditions, use of high-quality reagents, and proper handling techniques reinforce stability. Professionals must combine these measures with routine verification to overcome variables that calibration alone cannot address. A strategic approach to calibration and monitoring ensures solutions remain reliable under diverse laboratory or industrial conditions.
Case Study:
Maintaining pH stability in aqueous solutions demonstrates calibration’s effectiveness. Frequent measurements using a calibrated pH meter with a pH Calibrating Buffer show that drift occurs over time. By calibrating before each test, laboratories maintain consistent readings and prevent unwanted chemical reactions. This practice protects sensitive reagents, improves experimental reproducibility, and emphasizes the importance of precise calibration in maintaining solution integrity. Reliable instruments ensure scientists can replicate results and maintain confidence in their data across multiple experiments or production batches.
Conclusion:
Calibration is critical for maintaining solution stability. Accurate measurements prevent variability, safeguard experiments, and ensure compliance across industries. Using standardized tools, proper storage, and routine monitoring preserves solution integrity over time. Laboratories, manufacturers, and quality-driven sectors rely on calibration to achieve reproducible, high-quality outcomes. Systematic calibration routines safeguard chemical composition, reduce errors, and maintain operational efficiency. Maintaining stable solutions through careful calibration practices ensures consistent results, reliable experiments, and trust in both laboratory and industrial processes.
FAQs
How often should instruments be calibrated to maintain stability?
Frequency depends on the instrument, usage, and solution sensitivity. Daily, weekly, or monthly checks may be necessary for precise operations.
Can calibration prevent chemical degradation?
Calibration does not stop reactions but provides accurate monitoring to take corrective actions before solution properties are compromised.
Which tools are most important for solution calibration?
pH meters, conductivity probes, titration devices, and standardized buffers are essential for maintaining accurate measurements.
How does calibration benefit industrial production?
It stabilizes process conditions, improves product consistency, reduces waste, and maintains regulatory compliance in manufacturing.
What common errors affect solution stability despite calibration?
Using expired standards, improper handling, and neglecting instrument drift can compromise stability and produce inconsistent results.